The key villain in migraine finally brought to life
-
Date
Thu 13 Sep 18
Migraine is the third most common illness in the world and the sixth most disabling disease, affecting one billion people worldwide.
Now a major international collaboration involving the University of Essex has brought to life a key chemical process in the brain which can lead to migraine, which will help further the understanding and treatment of this debilitating condition.
The research, led by the Monash Institute of Pharmaceutical Sciences in Australia (MIPS), and published in the prestigious journal Nature reveals for the first time the molecular details of the structure and dynamics of one of the key initiators of a migraine headache.
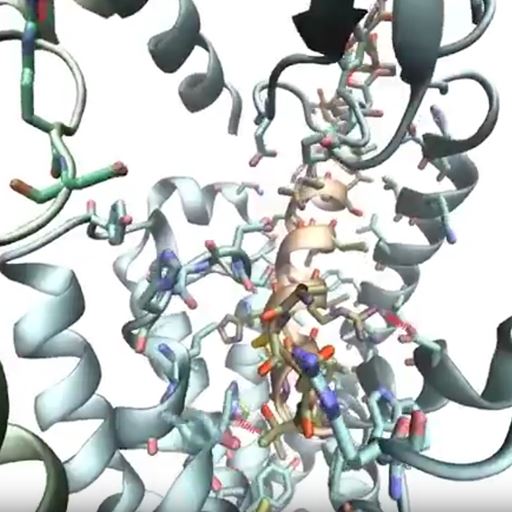
A key player in the initiation and pain of migraine is a signaling molecule called CGRP which works with the CGRP receptor, initiating a chain of events that can cause migraine. The research has presented the first high-resolution structure of this relationship.
Earlier this year the United States approved the use of Amovig - the first drug to prevent migraines by blocking the CGRP receptor – and several other such drugs are in the pipeline.
The dynamic structure developed in this multi-disciplinary, collaborative study will help further advance this area of drug design to treat and prevent migraines.
Scientists at Essex have been working on computational methods for simulating the structure and dynamics of this CGRP receptor for over seven years, and have been working with the Monash group for the last three years.
Professor Chris Reynolds, from Essex’s School of Life Sciences (formerly School of Biological Sciences), said: “We were privileged to work with colleagues elsewhere on this study, particularly Monash, who have been working for many years on getting this structure right. This partnership and the work of our brilliant post-doc Giuseppe Deganutti has brought this structure to life by showing the dynamics of how the CGRP receptor moves.”
Lead researchers, Professor Patrick Sexton and Dr Denise Wootten, of the Drug Discovery Biology theme at MIPS, said the study’s findings were a major breakthrough for understanding how selectivity of GPCRs could be controlled by novel protein partners.
“The CGRP receptor structure is the first example where we have been able capture a snapshot of a GPCR that has this complex mechanism of action,” said Professor Sexton.
“Excitingly, the CGRP receptor is not just a villain, but can also be activated for beneficial outcomes,” said Dr Wootten. “There is accumulating evidence that activation of the receptor could be used to treat inflammatory bowel disease or resistant hypertension.”
Driven by MIPS researchers including lead author Dr Lynn Liang, the research also involved the Max Planck Institute of Biochemistry in Germany, the University of Auckland in New Zealand, Fudan University in China and the Mayo Clinic in the US.




